Recently, the research team led by Professor Sun Mingjian from the School of Information Science and Engineering, in collaboration with the Shenzhen Institute of Advanced Technology of the Chinese Academy of Sciences and the Hong Kong Polytechnic University, has achieved significant progress in photoacoustic endoscopic imaging and safety monitoring for interventional hyperthermia therapy. The team proposed "a photoacoustic endoscopic temperature measurement strategy and system based on spatial adaptive weighting," which enables real-time, tissue-adaptive, and high-precision temperature monitoring inside blood vessels under interventional conditions. The relevant findings were published under the title "Real-Time, Tissue-Adaptive Photoacoustic Thermometry for Precision Endoscopic Thermal Therapy" in the top-tier optics journal Laser & Photonics Reviews.

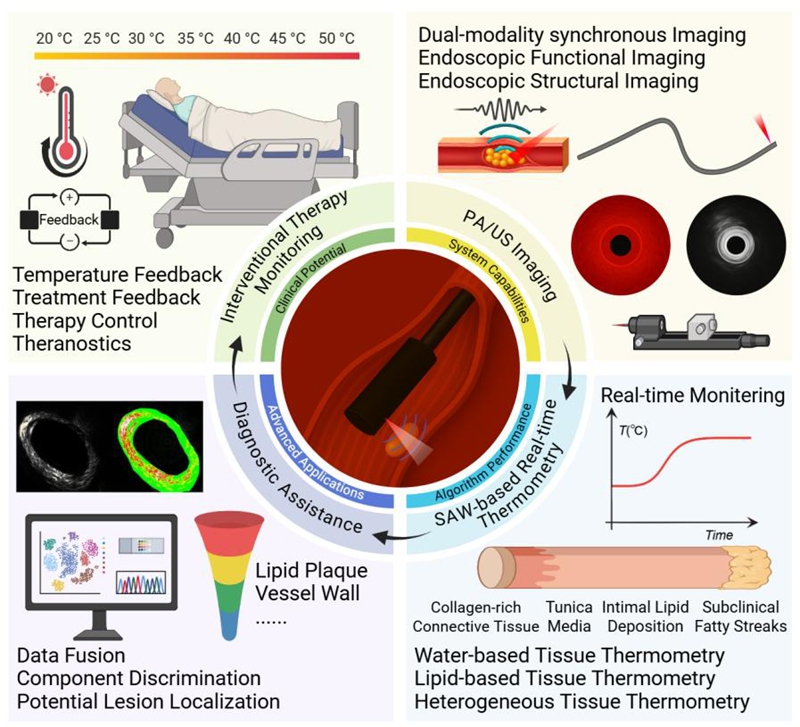
Atherosclerosis is a major cause of cardiovascular and cerebrovascular events, and the rupture of vulnerable plaques can induce serious consequences such as acute myocardial infarction and stroke. Intravascular photothermal ablation provides a new method for "precise reduction" of plaque volume, but the narrow and complex vascular lumen, the highly heterogeneous blood flow and tissue composition, and the continuous changes in the heated position caused by heartbeat and minute catheter displacements result in rapid temperature field changes, necessitating reliable thermal monitoring methods. Existing thermocouples and fiber optic probes are insufficient to accurately reflect local overall temperature changes, while multi-element systems struggle to effectively distinguish between deep vessels and plaque lesions. Achieving temperature monitoring that balances spatial resolution, temporal resolution, and clinical usability in the dynamic and complex vascular environment has become a significant bottleneck restricting the development of interventional hyperthermia.
To address this challenge, the research team independently developed a photoacoustic endoscopic imaging and temperature measurement system for vascular interventional scenarios and proposed a "spatial adaptive weighted" temperature measurement strategy. The system acquires photoacoustic images of the blood vessel cross-section within the catheter. The algorithm extracts angular features that are more sensitive to temperature changes from these images, distinguishing the different responses of water-based tissues and lipid plaques. Based on the signal-to-noise ratio and stability, the weights and calibration parameters are automatically adjusted, enabling the temperature measurement process to adapt to differences in tissue composition and improving the stability and reliability of the overall temperature readings.
The research team systematically validated the performance of PAET-SAW on three types of samples: water-based phantoms, lipid-based phantoms, and isolated rabbit abdominal aortas with atherosclerotic plaques. The results showed that, under both water bath and continuous laser heating modes, with real-time temperature monitoring at 20 frames per second, the maximum temperature measurement deviations in water-based and lipid-based phantoms were approximately 0.56℃ and 0.65℃, respectively. The temperature measurement error in isolated blood vessel samples was also controlled within 0.68℃, which is beneficial for early detection of potential overheating risks.
This research provides a novel solution for achieving high-real-time, high-precision intravascular temperature imaging in interventional photothermal therapy scenarios, offering strong support for real-time temperature monitoring and safety assessment in atherosclerotic plaque ablation, vascular closure, and other intravascular hyperthermic surgeries. In the future, the research team will further conduct in vivo animal and preclinical studies to systematically evaluate the stability and versatility of this strategy under complex blood flow and motion interference conditions, and explore its integration with multimodal imaging, closed-loop power modulation, and other technologies to accelerate the clinical translation of related findings.
Wu Dongjian, a doctoral student at the School of Information Science and Technology, is the first author of the paper. Professor Sun Mingjian, Professor Lai Puxiang of Hong Kong Polytechnic University, and Researcher Gong Xiaojing of the Shenzhen Institutes of Advanced Technology, Chinese Academy of Sciences, are the co-corresponding authors. Lecturer Ma Yiming, master's student Yu Kaicheng, and master's student Zhang Haokun from the research group made significant contributions to the publication of the paper. This research was supported by the National Key Research and Development Program of China (Key Project of Diagnostic Equipment and Biomedical Materials), the National Natural Science Foundation of China, the Shandong Provincial High-end Medical Device Innovation and Entrepreneurship Community Project (Technology Research and Development), and the Harbin Institute of Technology Young Scientist Studio. (Wu Dongjian)
Link to the Paper:https://doi.org/10.1002/lpor.202502499



